|
A liquid is more ordered than a gas, therefore entropy has decreased. This is as we would expect since we have gone from two gases to a gas and a liquid. Notice the negative sign, this indicates that entropy has decreased in the reaction. Calculate the entropy change for the reaction using the equation. 
You can work out the entropy of the reactants by adding up the entropy values for each reactant in a reactionģ. Entropy is a measure of how dispersed and random the energy and mass of a system are distributed. You can work out the entropy of the products by adding up the entropy values for each product in a reactionĢ. This is just the difference in entropy between the sum of the products and the sum of the reactants.Įxample: Calculate the entropy change for this reaction:ġ. 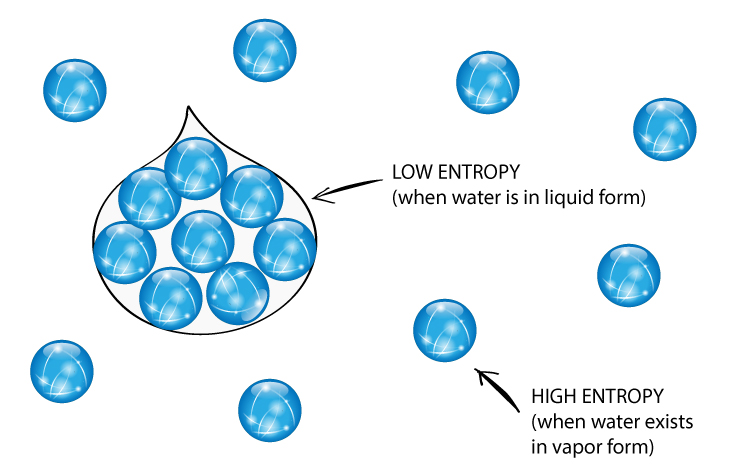
The formula for calculating ΔS is as follows: They are typically found at standard conditions. In a chemical reactions, products and reactants have different entropies – there is an entropy change.įor reactions with a negative entropy change, reactants will have a higher entropy than products.įor reactions with a positive entropy change, products will have a higher entropy than reactants.Įntropy values have been determined and can be found in databases. Answer: Since we start with equal amounts of water, the final temperature will be 22 ° C. Therefore, substance will naturally move in order to increase their entropy. The more disordered a system is, the higher its entropy and the more stable it is. When in a reaction the number of moles of product formed is greater than the number of moles of reactants, there is an increase in entropy. Number of particles – For an increased number of particles there is and increased number of ways in which these can be arranged and therefore an increased entropy. Dissolving – When a solid dissolves in a solvent, its entropy increases because of the free movement of the dissolved particles.ģ. Thermodynamic – Introduction to EntropyĢ. This is because the arrangement of particles is more random in gases than solids. State of Matter – Generally, solids have lower entropy than liquids, which have lower entropy than gases. The more disordered a system is, the higher (the more positive) the value of entropy.ġ. Send us feedback about these examples.Thermodynamic - Introduction to Entropy (A-Level Chemistry) Introduction to Entropy Entropy Key TermsĮntropy can be defined as the randomness or dispersal of energy of a system. These examples are programmatically compiled from various online sources to illustrate current usage of the word 'entropy.' Any opinions expressed in the examples do not represent those of Merriam-Webster or its editors. Sebastian Smee, Washington Post, After seven years in the Hammerskins, exhaustion and entropy were setting in. 2022 Succumbing to something closer to entropy than evolution, the watercolor starts to puddle and bruise. Ahmed Almheiri, Scientific American, 17 Aug. 
2021 This is the island formula for the entanglement entropy of the Hawking radiation. Conor Feehly, Discover Magazine, 3 Nov. James Riordon, Scientific American, In short, the tendency for systems to move from low entropy to high entropy, the particular spacetime conditions of our solar system and the indeterminacy of the future combine to create our particular conception of time. James Riordon, Scientific American, The expansion allows the universe to smooth out, dissipating the entropy before collapsing again. Quanta Magazine, That flaw is entropy, which builds up as a universe bounces. 2022 If the entropy of the system decreases, the entropy of the environment must increase such that the sum of the two entropies can only increase or stay the same, but never decrease. Jennifer Ouellette, Ars Technica, 2 Dec. Recent Examples on the Web Jacob Bekenstein realized in 1974 that black holes also have entropy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
